Inverse design of structural colours in polymeric films with crystallization-induced reversible thermochromism
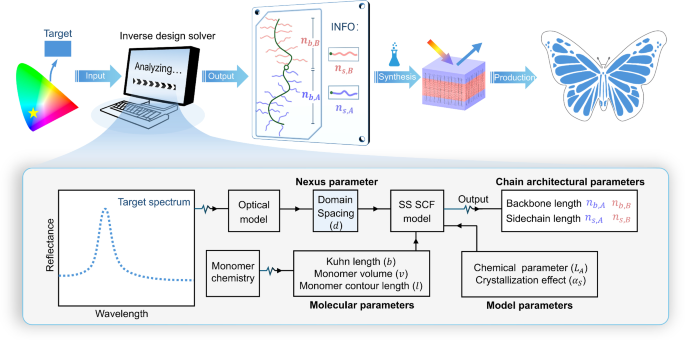
Colour design model
To avoid the conventional trial-and-error method, we aim to develop an inverse design approach to make structural colours in BBCPs. By inputting a target colour into an “inverse design solver”, we seek to directly obtain necessary side chain lengths (\({n}_{s,A},{n}_{s,B}\)), and backbone lengths (\({n}_{b,A},{n}_{b,B}\)), for given monomer chemistries (Fig. 1). The predicted structure is then synthesised and assembled to achieve the target colour, eliminating trial-and-error synthesis.
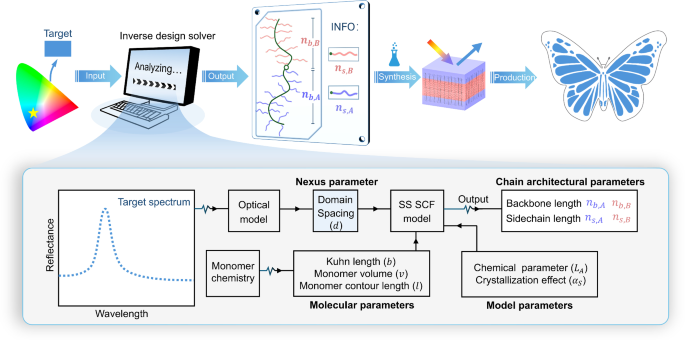
A target colour is input to an “inverse design solver” to obtain the required BBCP chain architectures for given monomer chemistries. The solver is a colour design model that integrates a multilayer optical model (relating spectra to domain sizes) and an SS-SCF model (bridging domain sizes to chain architectures) through a “nexus” parameter, namely domain spacing (\(d\)).
To construct the inverse design solver, we integrate a polymer physics model with an optical model to establish a direct link between molecular structures and macroscopic colours. The polymer physics model quantitatively maps molecular structures to nanostructures, while the optical model relates nanostructures to specific colours. The two models are coupled through a “nexus” parameter, domain spacing (\(d\)). Considering the multilayer structure is most common in bulk BBCPs, we adopt a multilayer optical model to obtain the best-fit spectrum from a target colour and then extract key parameters, namely domain spacing and refractive index. For given monomer chemistries, the refractive index is fixed, making the domain spacing the primary determinant of colour.
The theoretical domain spacings (\({d}_{{{\rm{thy}}}}\)) can be predicted from BBCP molecular structures using SS-SCF theory. Inspired by the framework of Zhulina et al. 33, we incorporate chemical disparities between backbone and side chains, which influence bottlebrush polymer conformation35. As shown in Fig. 1, for a given monomer chemistry, we first calculate molecular parameters including Kuhn length \((b)\), monomer volume (\(v\)), and monomer contour length (\(l\)) for both backbone and side chains. We then define a characteristic length scale, \({\widetilde{L}}_{A}\), which is a function of two measurable parameters, critical crowding parameters (\({\Phi }_{{{\rm{A}}}}^{*}\), \({\Phi }_{{{\rm{B}}}}^{*}\)) and interfacial tension (\({\gamma }_{A/B}\)) (details in Supplementary Section 1.1–1.3). For crystalisable side chains, we assume that crystallisation influences domain spacing through increasing domain density and inducing side chain stiffening, phenomenologically described by a side chain stiffening parameter, \({\alpha }_{s}\) (details in Supplementary Section 1.3). Using both molecular and model parameters (\({\widetilde{L}}_{A}\) and \({\alpha }_{s}\)), the domain spacing is computed following Eq. (26) in Supplementary Information.
We validate the SS-SCF model by comparing the predicted theoretical domain spacings \(({d}_{{{\rm{thy}}}})\) with the experimental domain spacings (\({d}_{{{\rm{expt}}}}\)). We choose PDMS-b-PEO as a model system due to its strong microphase separation, driven by a large Flory-Huggins parameter (\(\chi \approx\) 0.21)36, low glass transition temperature of PDMS (\({T}_{g}\) ~ −125 °C)37, and the crystallizability of PEO38. We expect PDMS and PEO blocks to undergo strong segregation to produce sharp interfaces and uniform self-assembly structures, thereby ensuring that real samples closely resemble those in the multilayer optical model. Using reported values for critical crowding parameter and interfacial tension of PDMS-b-PEO39, we calculate \({\widetilde{L}}_{{\mathrm{PDMS}}}=\) 0.44 nm (details in Supplementary Section 1.3). This allows us to use the SS-SCF model to determine the range of chain architectural parameters required to obtain lamellar structures with domain spacings of 150–300 nm at the melt state (without crystallisation) (Fig. 2a). Within this range, we synthesise four PDMS-b-PEO BBCPs via sequential ring opening metathesis polymerisation (ROMP)40, with different backbone degrees of polymerisation (Supplementary Figs. 3–8). BBCPs are assembled into bulk photonic films (>100 \({{\mu }}{{\rm{m}}}\)) by solution casting in a toluene atmosphere, followed by vacuum annealing at 100 °C for 8 h. (Fig. 2b, right). The toluene is used because it is a good solvent for most polymers, and its moderate evaporation rate offers enough time for polymer chains to undergo microphase separation to ordered structures during solution casting. Increasing block lengths (\({n}_{b,{{\rm{PDMS}}}}+{n}_{b,{{\rm{PEO}}}}\)), causes a blue-to-red colour shift, consistent with the expected redshift (Fig. 2c).
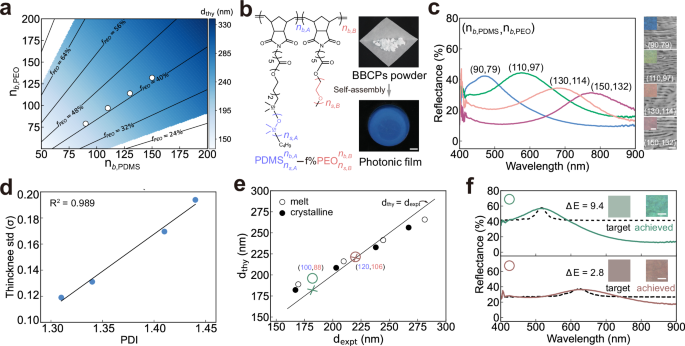
a Predicted domain spacings as a function of backbone lengths for fixed side chain lengths (\({n}_{s,{{\rm{PDMS}}}}=\) 68, \({n}_{s,{{\rm{PEO}}}}=\) 105) based on the SS-SCF model in the absence of crystallisation. The blue region represents the lamellar phase (L) and the white region represents other structures such as cylindrical (C, C’) or spherical (S, S’) phases. b Molecular structure of PDMS-b-PEO, along with images of the as-synthesised material and photonic film after self-assembly. Scale bar, 5 mm. c Reflectance spectra of photonic films with increasing molecular weights obtained by a microspectrometer. Insets are optical images and cross-sectional SEM images. Scale bars are 50 \({{\mu }}{{\rm{m}}}\) in optical images and 500 nm in SEM images. d Correlation between the optimal standard deviation of the layer thickness and BBCP polydispersity. e Comparison of theoretical and experimental domain spacings for four PDMS-b-PEO BBCPs at both melt (open circles) and crystallised (filled circles) states. Coloured crosses and circles are target and experimentally achieved domain spacings. f Reflectance spectra of target colours (dotted line) and corresponding experimentally achieved colours (solid line) using PDMS-b-PEO BBCPs with \({n}_{b,{{\rm{PDMS}}}}=\) 100 and \({n}_{b,{{\rm{PEO}}}}=\) 88, \({n}_{b,{{\rm{PDMS}}}}=\) 120 and \({n}_{b,{{\rm{PEO}}}}=\) 106.
From the measured reflectance spectra, we use a modified multilayer optical model to extract experimental domain spacing (\({d}_{{{\rm{expt}}}}\)). Cross-sectional SEM images reveal multilayer structures with some variations in layer thickness and orientations (insets in Fig. 2c). To account for these variations, we model the PDMS and PEO layer thickness with a truncated normal distribution and incident angles ranging from 0° to 24° (details in Supplementary Section 2). The means and standard deviations of the layer thickness distributions are determined through two steps of Bayesian optimisation, minimising colour differences between simulated and experimental spectra (Supplementary Figs. 2, 9). As shown in Fig. 2d, the standard deviation of layer thickness correlates linearly with the dispersity index (PDI) of PDMS-b-PEO. This suggests that this modified optical model can predict the thickness variations for each layer simply based on BBCP’s PDI.
These samples crystallise at room temperature, as confirmed by differential scanning calorimetry (DSC) spectra (Supplementary Fig. 10). To first validate the SS-SCF model in the absence of crystallisation, we measure the reflectance spectra at the melt state (\(T=\) 80 °C > \({T}_{m}=\) 57 °C, Supplementary Fig. 11) and extract the experimental domain spacing using the modified multilayered model. By fitting the calculated domain spacings to the experimental values at the melt state using Eq. (26) (details in Supplementary Section 1.3), we determine \({\widetilde{L}}_{{{\rm{PDMS}}}}=\) 0.44 nm, in excellent agreement with the theoretically calculated \({\widetilde{L}}_{{{\rm{PDMS}}}}=\) 0.44 nm. Incorporating a stiffening parameter of αs = 1.96 yields theoretical predictions that align well with experimental domain spacings across all four BBCPs at the crystallised state (Fig. 2e, Supplementary Fig. 12). With the SS-SCF model validated under both melt and crystallised conditions, we can now use it to predict chain architectural parameters for a given domain spacing.
By integrating the modified multilayer optical model and SS-SCF model, we obtain a colour design model for inverse design of structural colours. For a green colour with a reflectance peak at 520 nm, the required domain spacing is calculated as 195 nm by the optical model with determined average layer thickness and standard deviation of the thickness distribution. We then input the domain spacing to the SS-SCF model to determine the corresponding chain architectural parameters. Among the feasible solutions, we select a BBCP with \({n}_{s,{{\rm{PDMS}}}}=\) 68, \({n}_{s,{{\rm{PEO}}}}=105,{n}_{b,{{\rm{PDMS}}}}=100\), and \({n}_{b,{{\rm{PEO}}}}=\) 88. The synthesised BBCP (\({{\mbox{PDMS}}}_{68}^{100}-\)46%\({{\mbox{PEO}}}_{105}^{88}\)) assembles into a green film with a reflection wavelength of 516 nm, closely matching the target (Fig. 2f). Using the same side chain lengths, a red colour design yields a target domain spacing corresponding to \({n}_{b,{{\rm{PDMS}}}}=\) 120 and \({n}_{b,{{\rm{PEO}}}}=\) 106. The resulting film displays a red colour in agreement with the design, though with a slightly broadened spectrum.
To extend the model to BBCPs with different monomer chemistries, we choose PDMS-b-PCL due to PCL’s crystallinity, comparable to PEO (Fig. 3a). Since the colour design model is currently limited to 1D lamellar structures, we need to determine the boundary conditions for PDMS-b-PCL BBCPs to form lamellar structures. Taking PDMS-b-PCL with \({n}_{s,{{\rm{PDMS}}}}=68,{n}_{s,{{\rm{PCL}}}}=29\) for example, we can calculate that lamellar structures fall in the range of PCL volume fraction of 28–68% at crystalline state (Supplementary Fig. 13). Guided by this, we synthesise and assemble four PDMS-b-PCL BBCPs with fixed side chain lengths (\({n}_{s,{{\rm{PDMS}}}}=\) 68, \({n}_{s,{{\rm{PCL}}}}=\) 29) and varying backbone lengths (\({n}_{b,{{\rm{PDMS}}}}=\) 90\(-150,{n}_{b,{{\rm{PCL}}}}=115-190\)) (Supplementary Figs. 14, 15). The resulting films show colours from blue to yellow (Supplementary Fig. 16) after annealing under vacuum at 100 °C for 8 h. Extending the annealing time causes almost no additional colour change, indicating that the lamellar structure reaches a metastable state if it is not at equilibrium (Supplementary Fig. 17). In addition, photonic films cast from different solvents show similar colours and their spectra peak positions are close to each other (Supplementary Fig. 18). This suggests these solvents do not cause much difference in colours of PDMS-b-PCL films, in contrast to a recent report that solvent can largely modulate the morphology and colour of polystyrene-block– polylactic acid (PS-b-PLA) BBCP films41. This is likely because PDMS and PCL side chains have fast chain dynamics compared to PS and PLA side chains and kinetically trapped structures during solvent evaporation can be effectively erased after subsequent thermal annealing. The reproducibility of our experiments establishes the basis for reliable model-guided inverse design.
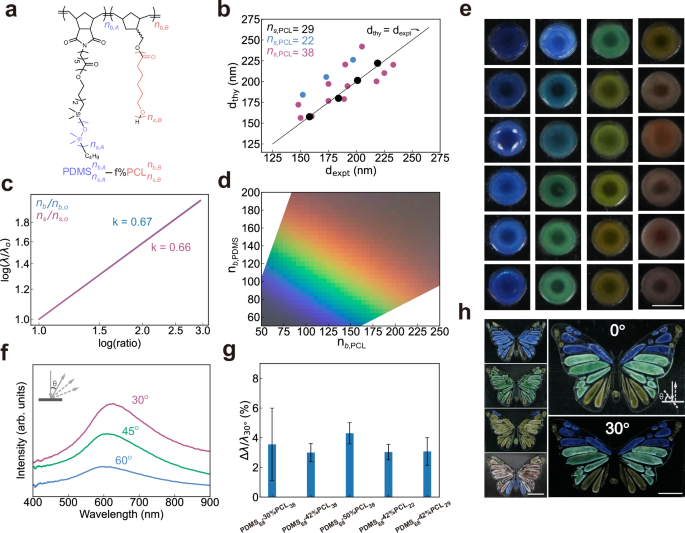
a Molecular structure of PDMS-b-PCL. b Comparison between theoretical and experimental domain spacings for PDMS-b-PCL BBCPs with different chain architectures (19 samples) at the crystallised state. Model parameters \({\widetilde{L}}_{A}\) and \({\alpha }_{s}\) are obtained by fitting four reference samples (black circles). c Effect of molecular architectural parameters on the reflectance peak with fixed side chain lengths (\({n}_{s,{{\rm{PDMS}}}}\)= 68, \({n}_{s,{{\rm{PCL}}}}\)= 38). d Predicted colour gamut from multilayer nanostructures when varying backbone lengths with fixed side chain lengths (\({n}_{s,{{\rm{PDMS}}}}\)= 68, \({n}_{s,{{\rm{PCL}}}}\)= 38). e Colour swatches produced by assembling BBCPs with various chain architectures. Scale bar, 1.5 mm. f Angle-resolved reflectance spectra of \({{\mbox{PDMS}}}_{68}^{150}\)−50%\({{\mbox{PCL}}}_{38}^{210}\) film, measured with normal incident illumination and detection angles from 30° to 60\(^\circ .\) g Reflectance peak shift ratios (\(\Delta \lambda /{\lambda }_{30^\circ }\)) for five groups of BBCPs with different molecular parameters. ∆\(\lambda={\lambda }_{30^\circ }-{\lambda }_{45^\circ }\) represents the peak shift when the detecting angle changes from 30° to 45° under fixed normal incident light. Data are presented as mean values \(\pm\) standard errors of the mean (n = 3 for \({{\mbox{PDMS}}}_{68}\)−30%\({{\mbox{PCL}}}_{38}\), \({{\mbox{PDMS}}}_{68}\)−50%\({{\mbox{PCL}}}_{38}\), \({{\mbox{PDMS}}}_{68}\)−42%\({{\mbox{PCL}}}_{22}\) and n = 4 for other samples). h Butterfly-shaped films displaying angle-dependent colours, guided by the colour design model. Scale bar, 5 mm.
To determine \({\widetilde{L}}_{{{\rm{A}}}}\) and \({\alpha }_{{{\rm{s}}}}\) for PDMS-b-PCL system, we run two steps of optimisation in the same manner as for the PDMS-b-PEO system. According to Eq. (26) in Supplementary Information, we first run a linear fitting using four measured layer spacings \(d\) in the melt state (obtained from reflection spectra in Supplementary Fig. 19a), combined with the polymer parameters listed in Supplementary Table 1. The slope gives \({\widetilde{L}}_{A}\) = 0.35 nm, which is then used to optimise \({\alpha }_{{{\rm{s}}}}\) based on layer spacing data in the crystallised state and gives \({\alpha }_{s}=\) 1.62 (black circles in Fig. 3b, Supplementary Fig. 19). This two-step optimisation approach ensures that each parameter retains a clear physical meaning while minimising the coupling between parameters.
To evaluate the model’s robustness across a broader design space, we then synthesise and assemble 22 more PDMS-b-PCL BBCPs with different side chains and volume fractions. The molecular weights range from 0.82 \(\times\) 106 to 3.28 \(\times\) 106 g/mol with PDI of 1.17–1.54 (Supplementary Fig. 20). All samples show lamellar structures, consistent to the prediction by the SS-SCF model (Supplementary Figs. 21–24 and Section 1.4). Only a few SEM images show lamellar structures mixed with local defects that are difficult to avoid for bulk films with thicknesses exceeding 100 μm. Experimental domain spacings, obtained from their reflectance spectra using the multilayer optical model, align well with the predicted spacings with these established \({\widetilde{L}}_{A}=\) 0.35 nm and \({\alpha }_{s}=\) 1.62 (Fig. 3b). We observe that predicted domain spacings are consistently larger than measured domain spacings for two sample groups (\({{\mbox{PDMS}}}_{68}\)−50%\({{\mbox{PCL}}}_{38}\) and \({{\mbox{PDMS}}}_{68}\)−42%\({{\mbox{PCL}}}_{22}\)), likely due to variation in crystallinity of different PCL side chain lengths (Supplementary Fig. 25). This deviation may also arise from experimental variations between different batches such as uncertainty in measuring small volumes of Grubb’s catalysts during synthesis. Therefore, these results support the model’s applicability across a broad parameter space, including backbone lengths, side chain lengths, and volume fractions.
We further use the colour design model to visualise how the chain architecture affects the macroscopic colours. As shown in Fig. 3c, the reflectance peak \((\lambda )\), which scales with domain spacing, follows a power-law dependence on both backbone and size chain lengths: \(\lambda \sim {({n}_{s,{{\rm{PDMS}}}}+{n}_{s,{{\rm{PCL}}}})}^{0.67}\) and \({\lambda \sim ({n}_{b,{{\rm{PDMS}}}}+{n}_{b,{{\rm{PCL}}}})}^{0.66}\). This trend offers design guidance for tuning colours in BBCPs. More importantly, we can use the model to generate an experimentally achievable colour space. For instance, by fixing \({n}_{s,{{\rm{PDMS}}}}\)= 68, \({n}_{s,{{\rm{PCL}}}}\)= 38, we can predict a full colour gamut by varying backbone lengths (Fig. 3d).
More BBCPs are synthesised and assembled to generate a full spectrum of colours (Fig. 3e, Supplementary Fig. 26). To demonstrate spatial patterning and practical applicability, four PDMS-b-PCL BBCPs are used to fabricate a butterfly-shaped coloured pattern (Fig. 3h). The resulting colours remain largely invariant with viewing angle, akin to the angle-independent behaviour of photonic glasses. Angle-resolved spectra confirm that colour changes are subtle with varying angles (Fig. 3f), with peak shifts as low as 3–4% across all samples with different chain architectures (Fig. 3g, Supplementary Fig. 27). This consistency suggests a uniform degree of structural disorder in these multilayer nanostructures, which contributes to their angular robustness.
Crystallisation-induced thermochromism
Beyond the static colour control, we next investigate the temperature-dependent optical response in these BBCPs films. As shown in Fig. 4a, a representative PDMS-b-PCL film turns from orange to brown as the temperature decreases from 60 °C to 0 °C, with a full colour recovery upon heating. The temperature-responsive colour change is reversible after at least 25 heating and cooling cycles (Fig. 4b). This thermochromic behaviour is also observed in 17 PDMS-b-PCL photonic films with other chain architectures (Supplementary Fig. 28), demonstrating the generality of this thermochromism. We use ultra-small angle X-ray scattering (USAXS) to examine structural changes in a representative photonic film after heating from 20 °C to 60 °C (Supplementary Fig. 29). In both states, we observe two scattering peaks with a q-value ratio of 1:2, suggesting the lamellar morphology retains after heating. However, the primary scattering peak shifts to lower q values, corresponding to an increase in the lamellar spacing (d) from 145.5 nm to 156.3 nm. These values are in excellent agreement with the values calculated from optical spectra based on Bragg’s law.
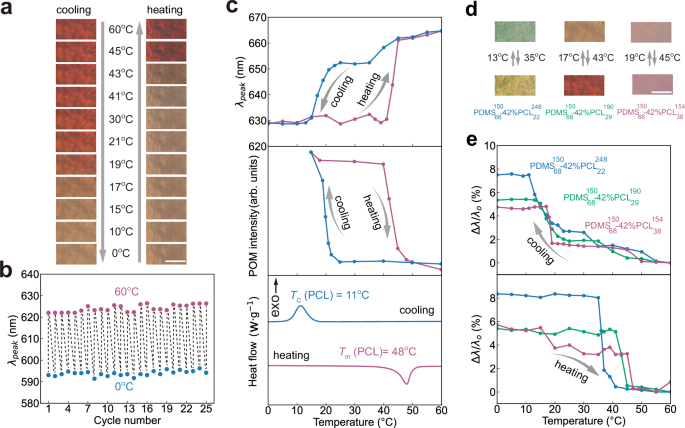
a Optical images of a PDMS-b-PCL film showing a reversible colour change between 0 °C and 60 °C during heating and cooling. Scale bar, 100 \({{\mu }}{{\rm{m}}}\). b Reflectance peak positions over 25 heating-cooling cycles, measured after equilibration at 0 °C and 60 °C. c Temperature-dependent evolution of reflectance peak wavelength, intensity of POM images, and normalised heat flow. The sample in (a, c) is produced by assembling \({{\mbox{PDMS}}}_{68}^{150}\)−42%\({{\mbox{PCL}}}_{29}^{190}\) and the sample in (b) is produced by assembling \({{\mbox{PDMS}}}_{68}^{130}\)−42%\({{\mbox{PCL}}}_{29}^{165}\). d Optical images of three PDMS-b-PCL samples at 0 °C and 60 °C with transition temperatures indicated. Scale bar, 100 \({{\mu }}{{\rm{m}}}\). e Percentage of reflectance peak shift during heating and cooling for three BBCPs: \({{\mbox{PDMS}}}_{68}^{150}\)−42%\({{\mbox{PCL}}}_{38}^{154}\) (red curve), \({{\mbox{PDMS}}}_{68}^{150}\)−42%\({{\mbox{PCL}}}_{29}^{190}\) (green curve), and \({{\mbox{PDMS}}}_{68}^{150}\)−42%\({{\mbox{PCL}}}_{22}^{248}\) (blue curve). All thermal scans are performed at a rate of 10 °C min−1.
To systematically explore the kinetics of colour change, we combine optical spectroscopy, polarised optical microscopy (POM), and DSC. In-situ reflectance spectra show a sharp spectral shift around 17 °C during cooling and around 43 °C during heating (Supplementary Fig. 30). The colour remains nearly unchanged after the temperature increases from 60 °C to 100 °C (Supplementary Fig. 31), suggesting a narrow thermochromic window. Plotting the peak shift with temperature reveals an asymmetric, nonlinear response during both thermal cycles (Fig. 4c). POM intensity plots show sharp transitions at melting (41 °C) and crystallisation (19 °C), closely matching the observed colour change (Supplementary Fig. 32). DSC measurements further confirm the melting (\({T}_{m}\) = 48 °C) and crystallisation (\({T}_{c}\) = 11 °C) of PCL side chains, consistent with colour change. These results collectively demonstrate that the colour change in photonic films originates from the phase transition in crystalline PCL blocks.
The colour transition temperature depends on the PCL side chain length. As \({n}_{s,{{\rm{PCL}}}}\) increases from 22 to 29 and 38, the colour change temperature rises from 13 °C to 19 °C during cooling and from 35 °C to 45 °C during heating (Fig. 4d, e). This trend aligns with DSC results of both homo bottlebrush PCL and PDMS-b-PCL with varying PCL chain lengths (Supplementary Figs. 33, 34). The elevated transition temperature observed in longer PCL chain lengths likely arises from stronger intermolecular interactions and the formation of extensive crystalline domains, which require higher thermal energy to melt. These findings show that we can tailor the colour-changing temperature simply by adjusting the PCL side chain lengths.
To quantitatively reveal the leading mechanism behind colour changes, we track the temperature-dependent evolution of film thickness and refractive index using a spectroscopic ellipsometer. As the temperature increases from 0 °C to 60 °C, the film thickness expands by 7.6%, while the refractive index at 589 nm decreases by 2.5% from 1.472 to 1.436 (Fig. 5a, b, Supplementary Fig. 35). Upon cooling, both the thickness and the refractive index recover with similar hysteresis to the melting and crystallisation transition of semicrystalline polymers. Using the measured temperature-dependent film thickness and refractive index, we apply Bragg’s law to calculate the shift in reflectance peak positions (Eqs. 35–37 in Supplementary Section 4). The calculated peak shift not only captures the trend observed in experimental spectra, but also quantitatively agrees with the measured shift: a predicted change of 5.0% matches the measured 5.6% from 60 °C to 0 °C (Supplementary Fig. 36). This consistency extends across PDMS-b-PCL samples with different molecular architectures (Supplementary Fig. 37).
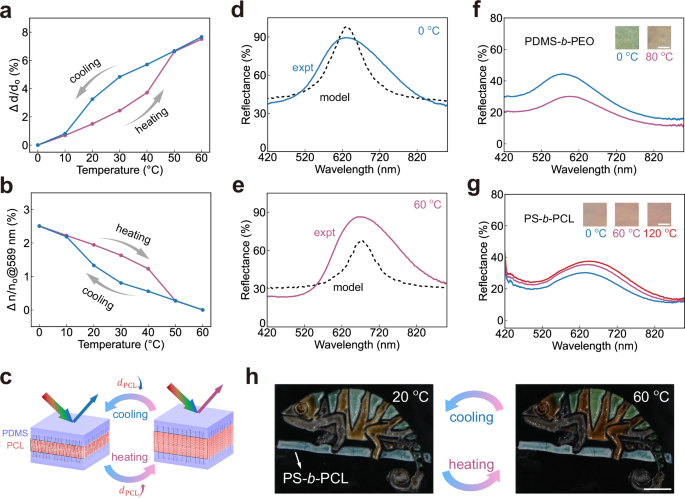
a, b Temperature-dependent changes in (a) refractive index and (b) thickness during heating and cooling cycles. Each data point is measured within 20 s at the corresponding temperature. The sample is assembled from \({{\mbox{PDMS}}}_{68}^{150}\)−42%\({{\mbox{PCL}}}_{29}^{190}\). c Schematic illustration showing domain spacing variations during thermal cycles. d, e Comparisons between experimental reflectance spectra taken by a microspectrometer with optical model predictions at 0 °C and 60 °C. f, g Reflectance spectra taken by a microspectrometer and optical images at different temperatures for (f) \({{\mbox{PDMS}}}_{68}^{110}\)−46%\({{\mbox{PEO}}}_{105}^{97}\) and (g) \({{\mbox{PS}}}_{37}^{110}\)−40%\({{\mbox{PCL}}}_{22}^{113}\) films. Scale bars: 50 \({{\mu }}{{\rm{m}}}\). h Chameleon-shaped pattern composed of thermochromic PDMS-b-PCL regions (body) with non-responsive PS-b-PCL (tree trunk), demonstrating selective colour change upon heating and cooling. Scale bar, 5 mm.
The PCL layer is expected to expand or shrink significantly more than the PDMS layer during thermal cycles due to its melting and crystallisation (Fig. 5c). To validate this, we further use the modified multilayer model to extract the layer thickness at both melt and crystallised states. Using the refractive indices of PCL and PDMS from ellipsometry measurements (details in Supplementary Section 4)42, we find that the simulated and experimental spectra align when the PCL layer increases by 15.9% and the PDMS layer increases by 0.9% upon heating from 0 °C to 60 °C (Fig. 5d, e). This suggests that the PCL thickness variation is the dominant contributor to the colour change. Interestingly, the PCL homo bottlebrush film expands only by 5.5% in thickness, almost three times less than the thickness expansion of the PCL layer in the PDMS-b-PCL film after melting (Supplementary Fig. 38). This demonstrates that PCL crystallisation causes both volume shrinkage and side chain stiffening, captured by the stiffening parameter (\({\alpha }_{s}\)) in the SS-SCF model.
We examine other semicrystalline BBCPs to assess the universality of thermochromism. A PDMS-b-PEO film exhibits a colour shift upon heating to 80 °C, exceeding the PEO’s melting temperature (\({T}_{m}=\) 57 °C) (Fig. 5f). In contrast, a coloured PS-b-PCL film shows negligible colour change even at 120 °C (Fig. 5g, chemical characterisations in Supplementary Figs. 7, 39). DSC analysis shows that the high glass transition temperature of PS (\({T}_{g}=\) 102 °C) restricts PCL crystallisation43, resulting in minimal colour change (Supplementary Fig. 40). PDMS-b-PCL and PDMS-b-PEO show significant crystallisation, likely facilitated by the low-\({T}_{g}\) PDMS block. These results demonstrate that BBCPs comprising a crystalline block with a soft, low-\({T}_{g}\) block enable temperature-responsive photonic films. Leveraging this thermochromic property, we design a chameleon-inspired pattern with a temperature-sensitive PDMS-b-PCL body and a non-responsive PS-b-PCL tree trunk. When heating from 20 °C to 60 °C, the chameleon’s blue and yellow stripes turn to green and orange, while the tree trunk remains unchanged. This suggests different BBCPs can be selected to achieve spatially tunable thermochromic patterning.
link






